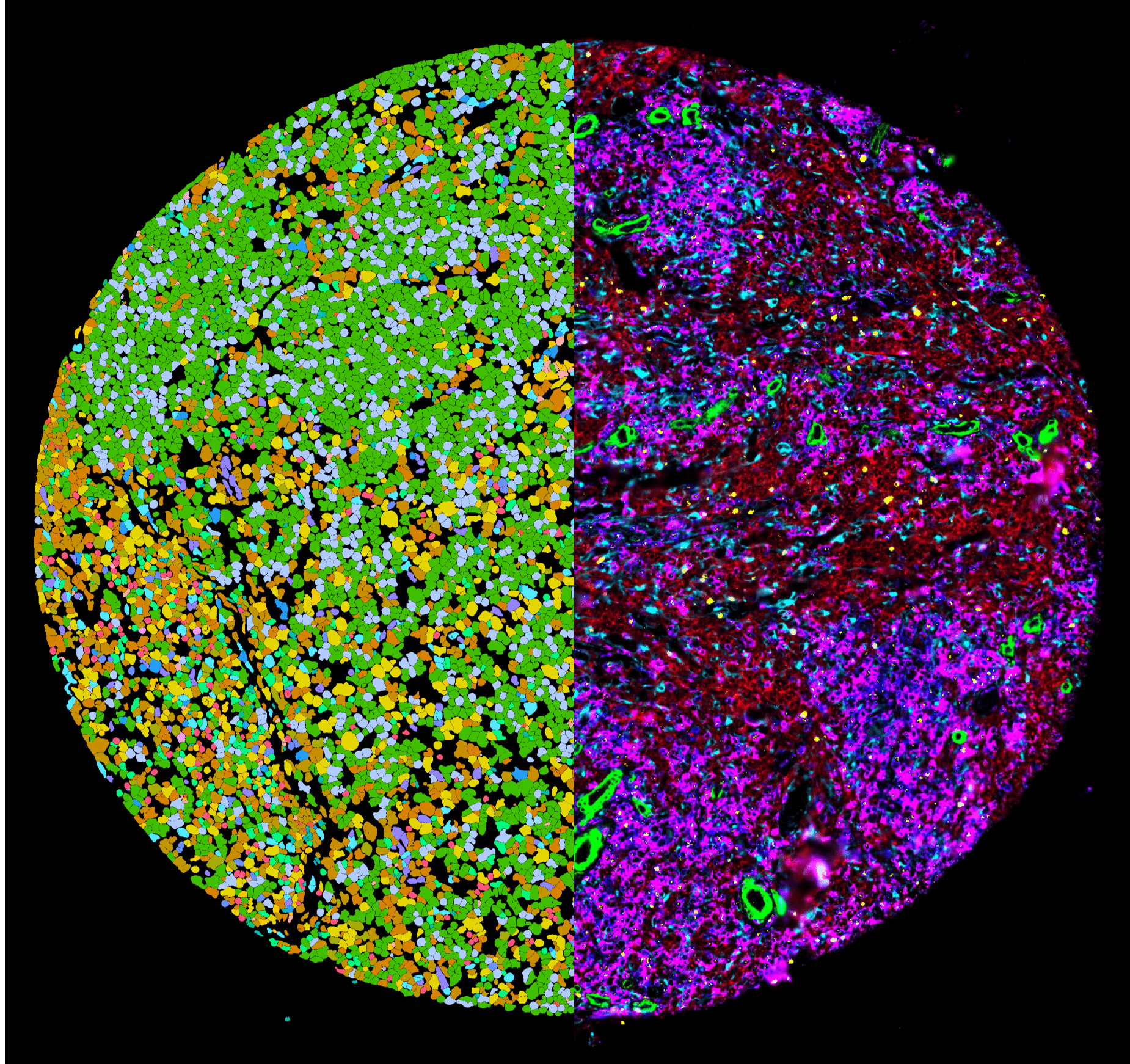Keywords
Lymphoma, prediction of response, spatial organization; liquid biopsy, individualized dynamic risk.
Overarching Goal/Mission Statment
Intercept3.0 aims to develop a dynamic, individualized risk and response prediction for lymphoid malignancies in adults by integrating longitudinal, multi-modal tumor and immune profiling before and early during treatment. This approach enables early detection of insufficient therapy response, allowing timely adaptation of treatment to improve patient outcomes, reduce unnecessary side effects, and maximize the chances of successful retreatment.
Aims
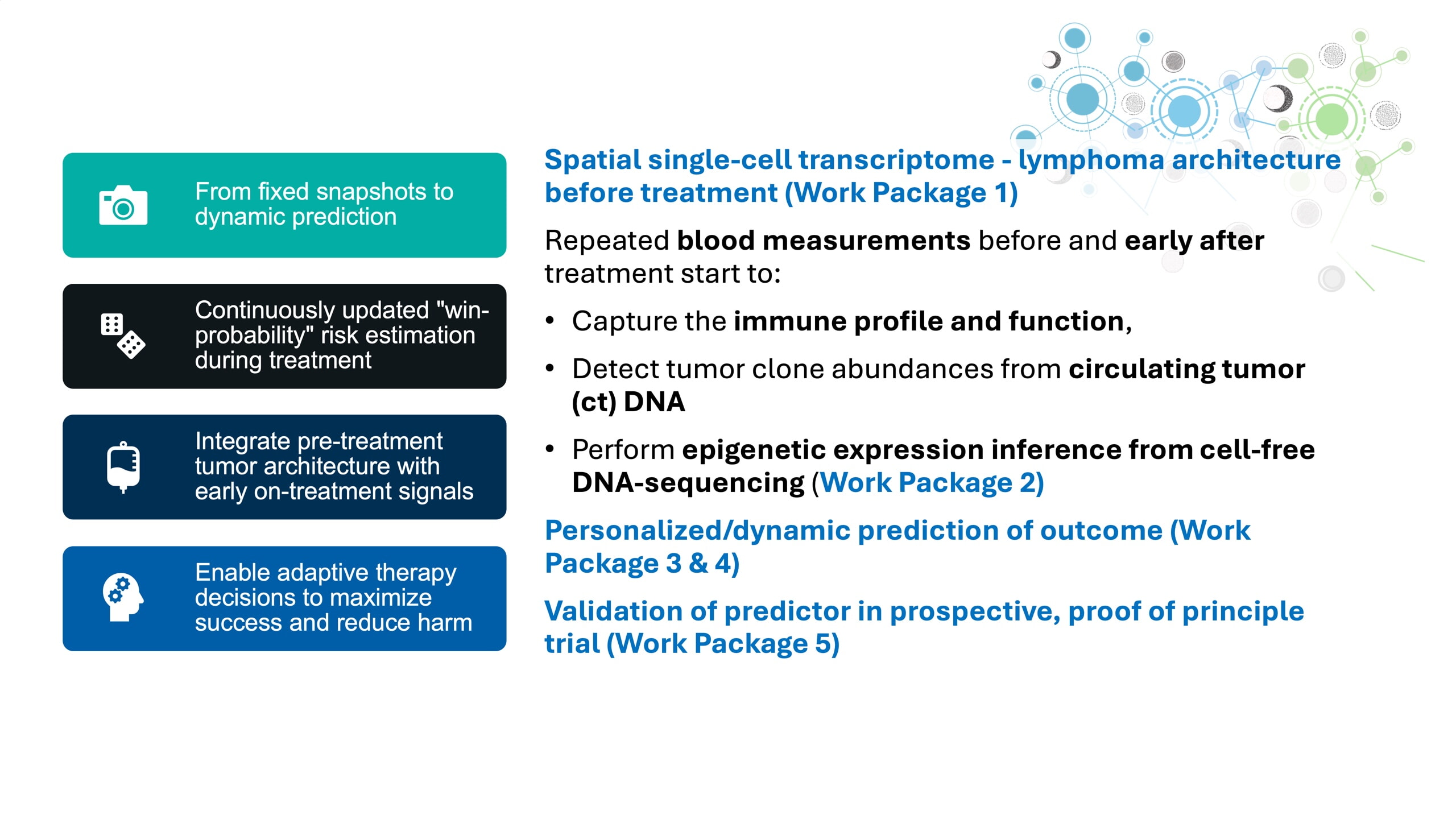
- Aim 1: Capture spatial single-cell landscape of lymphoma and develop approaches to predict tissue tumor architecture from blood
- Aim 2: Measure drug response in lymphoma by longitudinal measurements from blood
- Aim 3: Build computational models to predict patient treatment response based on multi-omic pre-treatment profiles and early measurements after treatment
- Aim 4: Demonstrate the feasibility of dynamic risk prediction in lymphoma in a prospective clinical trial
“We aim to predict the course of lymphoma patients as early as possible, using high-resolution analyses before and immediately after treatment. We hope these efforts will represent an important step toward truly personalized cancer therapy.”
Prof. Dr. med. Thorsten Zenz - Department of Medical Oncology and Hematology, USZ

Project Summary
Precision oncology tools often focus on a priori prediction of risk or response, i.e., based on biomarkers of specimens collected before treatment. To improve the outcome of lymphoma patients, we aim to improve precision medicine by a new, currently not available approach. Our ongoing multi-omics studies, which include longitudinal observations and measurements over years, put us in the unique position to combine multi-modal data on tumor composition before treatment with early, multi-modal measurements on/shortly after treatment to detect insufficient response and thus maximize opportunities for successful retreatment. We focus on common and often lethal lymphoid malignancies in adults (non-Hodgkin`s Lymphoma). We are developing a dynamic individualized risk prediction for lymphoma and its treatment, similar to ‘‘win probability’’ models in other fields. This transition would move the field from fixed-interval (imaging) to dynamic individualized response assessments already at earlier time points. We hypothesize that markers for such a precise adaptive risk assessment in individual patient trajectories can be discovered by combining highly-resolved and spatial pre-treatment assessments with early —on treatment— measurements. We make use of our ongoing characterization of samples and clinical data from multiple cohorts and a feasibility trial (INTeRCePT2.0), where a total of up to 1.000 samples have already been analyzed, including patients with cellular therapy (CAR-T), bispecific antibodies, and chemoimmunotherapy. In a well-structured program, we now perform spatial single-cell transcriptome profiling of lymphoma and integrate these data with existing bulk and single-cell multi-omics profiles generated in prior LOOP projects (n=500 patients) to resolve tumor composition and architecture before treatment. We use repeated blood measurements before and early after treatment start to 1) capture the immune profile and function by high-dimensional cytometry, 2) detect tumor mutations from circulating tumor (ct) DNA, and 3) perform epigenetic expression inference from cell-free DNA-sequencing (EPIC-seq). We develop quantitative mathematical models and use machine learning to integrate different modalities and derive a personalized prediction of outcome. We then validate this predictor in a subsequent prospective trial (proof of principle). This technologically advanced, dynamic multi-modal strategy distinguishes our proposal fundamentally from other approaches. Early detection of resistance will spare patients treatments (and side effects) and maximize the opportunity to find active treatment(s), by shorter exposure to drugs that do not work.
Principal Investigator and Project Team

Prof. Dr. med. Thorsten Zenz, University Hospital Zurich, Department of Medical Oncology and Hematology, thorsten.zenz@usz.ch

Prof. Dr. Burkhard Becher, University of Zurich, Institute of Experimental Immunology, becher@immunology.uzh.ch

Prof. Dr. Niko Beerenwinkel, ETH Zurich, Institute for Biomedical Engineering, niko.beerenwinkel@bsse.ethz.ch

Prof. Dr. Valentina Boeva, ETH Zurich, Department of Computer Science, valentina.boeva@inf.ethz.ch

Dr. med. Marco M Bühler, University Hospital Zurich, Department of Pathology and Molecular Pathology, marcomatteo.buehler@usz.ch

Prof. Dr. Wolfgang Huber, EMBL, wolfgang.huber@embl.org

Prof. Dr. Andreas Moor, ETH Zurich, Department of Biosystems Science and Engineering , andreas.moor@bsse.ethz.ch

Prof. Dr. Virginie Uhlmann, University of Zurich, Department of Molecular Life Sciences, virginie.uhlmann@mls.uzh.ch
Funding Period
1.1.2026-31.12.2030
Funding Volume
CHF 4.050.000
Funded by
University Medicine Zurich (UMZH)
Comprehensive Cancer Center Zurich (CCCZ)
The LOOP Zurich
Tumor Profiler Center
Further projects
CCCZ Investigator Initiated Trial
BRAVE-Lung
Robotic-assisted Bronchoscopic Radiofrequency Ablation for Very-Early Lung Cancer -A novel Cancer Therapy
CCCZ Leuchtturmprojekte
Interdisziplinäre Forschungsteams übertragen Erkenntnisse aus der Grundlagenforschung in die klinische Anwendung und entwickeln neue Behandlungskonzepte gegen Krebs.