Keywords
Robotic-assisted bronchoscopy; minimally invasive therapy, radiofrequency ablation
Overarching Goal/Mission Statment
The goal of this project is to establish a safe, minimally invasive bronchoscopic therapy that combines diagnosis and treatment of early-stage lung cancer within a single procedure. By translating robotic bronchoscopy with Cone-Beam CT into a therapeutic platform, the study aims to create a new curative option for patients who are not candidates for surgery and to fundamentally improve early lung cancer care.
Aims
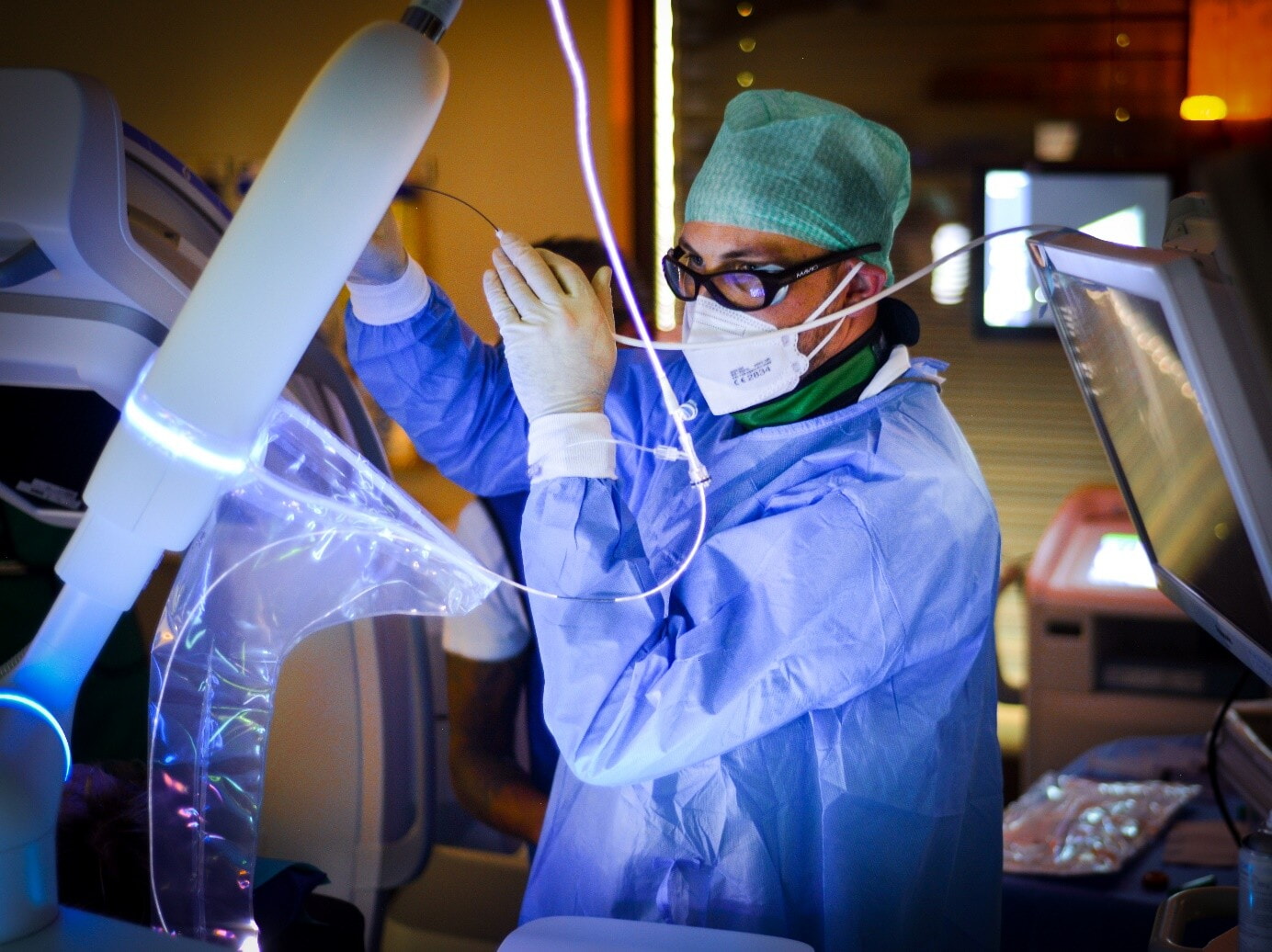
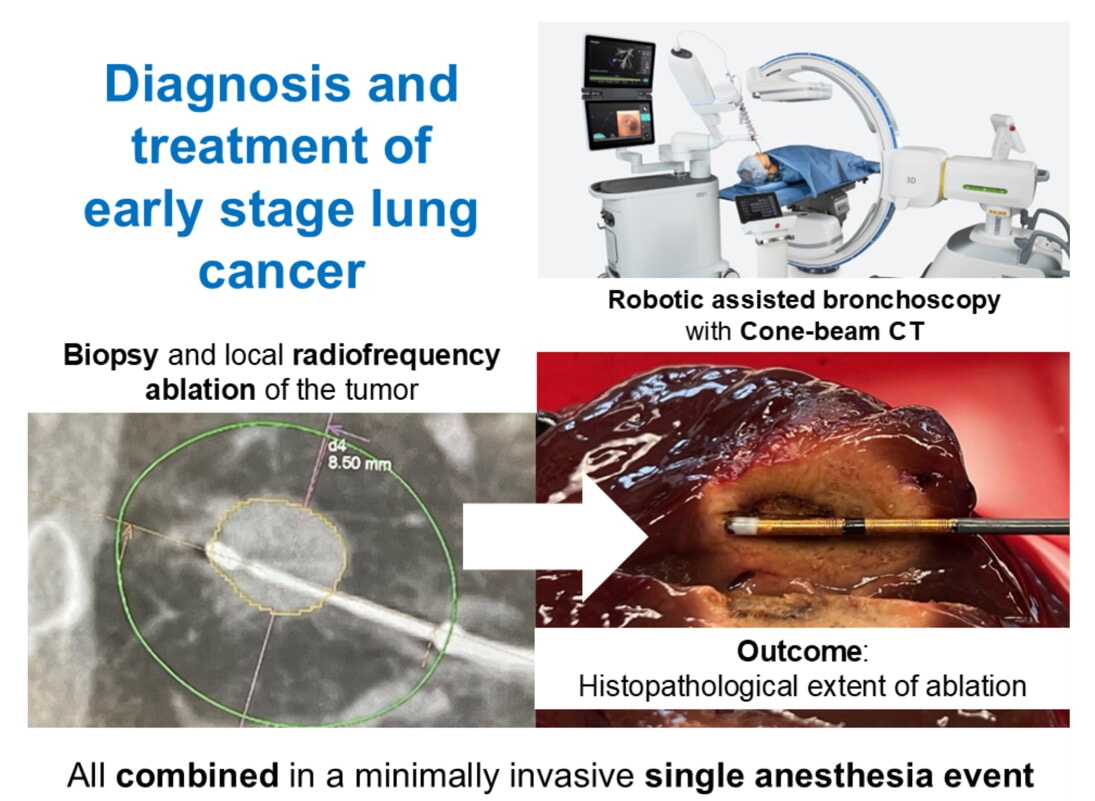
The objective of this prospective trial is to assess the procedural feasibility and post-resection histopathological effects of endobronchial radiofrequency-ablation (RFA) delivered via robotic-assisted bronchoscopy (RAB) with integrated cone-beam computed tomography (CBCT), embedded within the standard diagnostic bronchoscopy and surgical resection workflow in patients with histologically confirmed lung cancer. We hypothesize that combining high-precision RAB-CBCT navigation with an established ablation technology will enable effective local tumor control, thereby establishing endobronchial ablation as a feasible minimally invasive alternative for patients with early-stage non-small cell lung cancer (NSCLC). All interventions, including patient selection, tumor localization, and resection, follow current clinical standards. The RFA device is applied within its indication, and no randomization or deviation from standard of care is introduced.
“Our project introduces a new form of innovation in the treatment of very early-stage lung cancer by combining diagnosis and therapy into a single, minimally invasive procedure. Patients benefit from a gentler treatment, reduced risks, and the opportunity for earlier and more effective therapy.”
PD Dr. Thomas Gaisl PhD - Klinik für Pneumologie, USZ

Project Summary
Robotic-assisted bronchoscopy with integrated cone-beam computed tomography (RAB-CBCT) and cryobiopsies have transformed the diagnostic yield for peripheral pulmonary lesions by enabling high-precision, minimally invasive trans-parenchymal access. Since its implementation at the CCCZ, overall lung cancer diagnoses have increased by 69%, driven by a 291% increase in stage IA detections. These results suggest a potential paradigm shift in early-stage lung cancer management. The present first-in-human, prospective, multicenter study will evaluate whether this same technology platform can be safely and effectively extended from diagnosis to local treatment of stage IA non-small cell lung cancer (NSCLC) using a novel radiofrequency-ablation (RFA) probe. The primary aim is to determine the histopathological efficacy of bronchoscopic RFA in achieving complete tumor necrosis. Secondary aims include evaluating procedural safety, technical success (intra-procedural CBCT-confirmed ablation coverage), perioperative complications, and ablation-related tissue effects. The trial will enroll 40 patients with histologically confirmed or radiologically suspected stage IA NSCLC who are scheduled for curative-intent surgical resection. In a “treat-and-resect” design, patients will undergo RAB-guided diagnosis and RFA of a stage IA NSCLC (in a single anesthesia event) two weeks prior to surgery, allowing for direct histopathological assessment of ablation success in the resected specimens. Follow-up will extend to six weeks post-surgery and include clinical assessment, imaging, and pathological review. Statistical analysis will be descriptive, focusing on feasibility, safety, and histopathological outcomes in both the intention-to-treat and modified intention-to-treat cohorts. This design allows robust evaluation of a minimally invasive bronchoscopic treatment modality within the controlled setting of planned surgical resection. If successful, this study could establish local RFA as a stand-alone therapeutic option, enabling a singlesession diagnostic and treatment pathway for early-stage NSCLC. This approach would be particularly valuable for patients unfit for surgery and could redefine the standard of care in this population, analogous to curative strategies in other stage IA cancers such as esophageal cancer.
Principal Investigator and Project Team

Dr. med. Carolin Steinack, Leitende Ärztin, University Hospital Zurich, Department of Pulmonology, carolin.steinack@usz.ch

PD Dr. med. Thomas Gaisl, University Hospital Zurich, Department of Pulmonology, thomas.gaisl@usz.ch

Prof. Dr. med. Isabelle Schmitt-Opitz, University Hospital Zurich, Department of Thoracic Surgery, isabelle.schmitt-opitz@usz.ch

Dr. med. Martina Haberecker, University Hospital Zurich, Department of Pathology and Molecular Pathology, martina.haberecker@usz.ch
Funding Period
1.1.2026-31.12.2029
Funding Volume
CHF 445.000
Funded by
University Medicine Zurich (UMZH)
Comprehensive Cancer Center Zurich (CCCZ)
Further projects
Zurich Precision Oncology Consortium
INTeRCePT3.0
Dynamic individualized risk modeling to improve lymphoma treatment
CCCZ Leuchtturmprojekte
Interdisziplinäre Forschungsteams übertragen Erkenntnisse aus der Grundlagenforschung in die klinische Anwendung und entwickeln neue Behandlungskonzepte gegen Krebs.