Overarching Goal/Mission Statment
This project investigates the safety, preliminary efficacy and immunological effects of a novel cell therapy concept for patients with progressive glioblastoma after standard of care therapy. This is based on intratumorally administered, rapidly produced multifunctional CAR PBMCs that co-express pro-inflammatory cytokines.
Keywords
Glioblastoma, Immunotherapy, CAR, Phase 1
Aims
The primary objectives are to evaluate safety and to establish the recommended phase 2 dose of intratumorally administered multifunctional mRNA-based CAR-PBMCs in patients with recurrent glioblastoma who are eligible for another surgery after standard of care therapy. Secondary objectives include assessing preliminary therapeutic activity and immunological effects. A comprehensive translational research program, conducted in partnership with the C3Z Sample Processing Lab, will analyze CAR PBMC composition and characterize both cellular and humoral immune landscapes before and after therapy, using high-dimensional profiling of blood, tumor fluid, and, where available, tumor tissue specimens. We hypothesize that autologous multifunctional mRNA-based CAR PBMCs can be rapidly manufactured and safely administered intratumorally, where they will induce measurable immunostimulatory changes within the glioblastoma microenvironment, thereby promoting robust anti-tumor immune responses.
“This study gives patients access to the world’s first clinical trial using multifunctional CAR immune cells for refractory glioblastoma.”
PD Dr. Tobias Weiss - Department of Neurology, University Hospital Zurich (USZ)

Project Summary
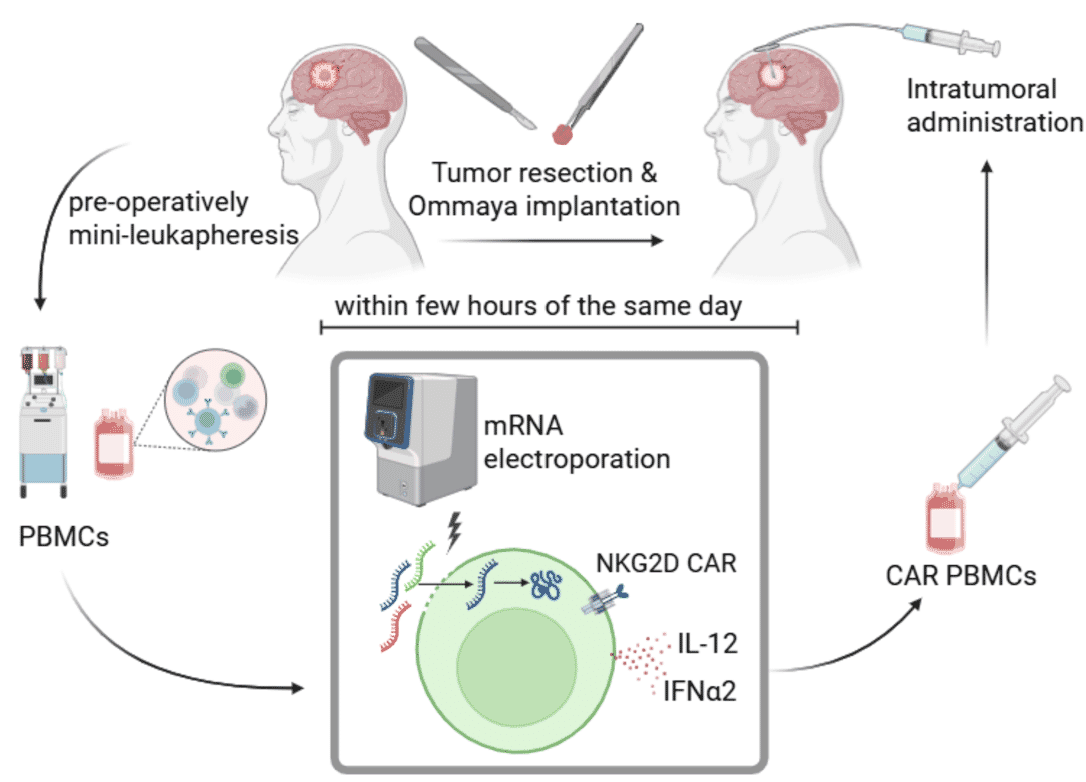
Glioblastoma is the most common and aggressive primary brain tumor in adults, with a dismal prognosis and lack of effective treatment options. Chimeric antigen receptor (CAR) immune cell therapy is a promising approach and early clinical trials have shown safety of locally administered CAR T cells in patients with glioblastoma but revealed key limitations of current approaches that hinder breakthrough: 1. limited efficacy because current approaches do not overcome the immunosuppressive tumor microenvironment and 2. current manufacturing processes are costly and take weeks, limiting widespread implementation and excluding many patients who progress before therapy initiation. We developed an innovative platform to overcome these hurdles. This platform uses modern mRNA technology to produce multifunctional CAR PBMCs within hours that express a multi-targeting CAR and pro-inflammatory cytokines, enabling same-day treatment and triggering a long-lasting bystander anti-tumor immune response. Our preclinical research demonstrates promising safety and potent anti-tumor immunity with a durable bystander anti-tumor immune response, and we established the clinical GMP-grade manufacturing platform within the scope of a former CCCZ lighthouse project. Based on the high medical need, promising preclinical data, the established manufacturing platform and positive interactions with the regulatory authorities, we now want to investigate the safety, preliminary efficacy and immunological effects of locally administered multifunctional CAR PBMCs in patients with recurrent glioblastoma in a first-in-the-world phase 1 monocenter, single-arm, open-label trial. PBMCs will be collected by short leukapheresis and electroporated with three mRNAs (NKG2D CAR, IL-12, IFN-α2) in a closed system without further cell culture. During surgery, an Ommaya reservoir and catheter will be placed in the tumor cavity for local CAR PBMC administrations. Patients will receive two infusions: immediately postsurgery and two weeks later. The trial will follow a 3+3 dose-escalation design, enrolling a minimum of 9 and a maximum of 18 patients if relevant toxicities occur. The primary objective is to evaluate safety and establish the recommended phase 2 dose, with endpoints including assessment of adverse events (AEs), serious adverse events (SAEs), dose-limiting toxicities (DLT) and the maximum tolerated / recommended phase 2 dose. The secondary objective is to assess preliminary efficacy and immunological effects, with endpoints such as progression-free survival and overall survival and accompanying translational immunomonitoring of blood, reservoir fluid and tumor tissue. We also assess quality of life measures using validated instruments. In summary, this phase 1 therapeutic trial addresses the unmet need for novel strategies in glioblastoma and has the potential to revolutionize cellular cancer immunotherapy by providing a widely implementable more costeffective, safer, and potentially more effective cell therapy approach.
Principal Investigator and Project Team

PD Dr. med. Dr. sc. nat. Tobias Weiss, Leitender Oberarzt, University Hospital Zurich, Department of Neurology, tobias.weiss@usz.ch

Prof. Dr. Dr. Dominik Schneidawind, University Hospital Zurich, Department of Medical Oncology and Hematology, dominik.schneidawind@usz.ch

PD Dr. med. Carlo Serra, University Hospital Zurich, Department of Neurosurgery, carlo.serra@usz.ch

Dr. med Stefanie Kreutmair, University Hospital Zurich, Department of Medical Oncology and Hematology, stefanie.kreutmair@usz.ch
Funding Period
1.1.2026-31.12.2029
Funding Volume
CHF 2.079.500
Funded by
University Medicine Zurich (UMZH)
Comprehensive Cancer Center Zurich (CCCZ)
Further projects
CCCZ Investigator Initiated Trial
BRAVE-Lung
Robotic-assisted Bronchoscopic Radiofrequency Ablation for Very-Early Lung Cancer -A novel Cancer Therapy
CCCZ Leuchtturmprojekte
Interdisziplinäre Forschungsteams übertragen Erkenntnisse aus der Grundlagenforschung in die klinische Anwendung und entwickeln neue Behandlungskonzepte gegen Krebs.