Aims
The goal of the project is to establish and apply two comple-mentary, innovative next-generation clinical CAR immune cell platforms which rely on virus-free technology. This will be accompanied by research on CAR immunomonitoring and efficacy improvement.
Keywords
Cancer immunotherapy, Chimeric antigen receptor, Non-viral gene transfer
Project Overview
CAR T cell immunotherapy is a new perspective of precision medicine which use gene transfer technology to generate T cells with improved anti-tumor activity. Despite their success in B-cell neoplasm, efficacy of CAR T-cell therapy is still limited in myeloid and solid tumors due to immunosuppressive tumor microenvironment, heterogeneous diseases, and poor tumor infiltration. Furthermore, most CAR T-cell therapies depend on viral vectors, which are associated with logistical complexity and high costs. The goal of the project is to establish and apply two comple-mentary, innovative next-generation clinical CAR immune cell platforms which rely on virus-free technology. This will be accompanied by research on CAR immunomonitoring and efficacy improvement.
Four questions to the principal investigator Chiara Magnani
Work Packages
1: A phase I clinical trial with locally administered multifunctional mRNA-based CAR immune cells in glioblastoma
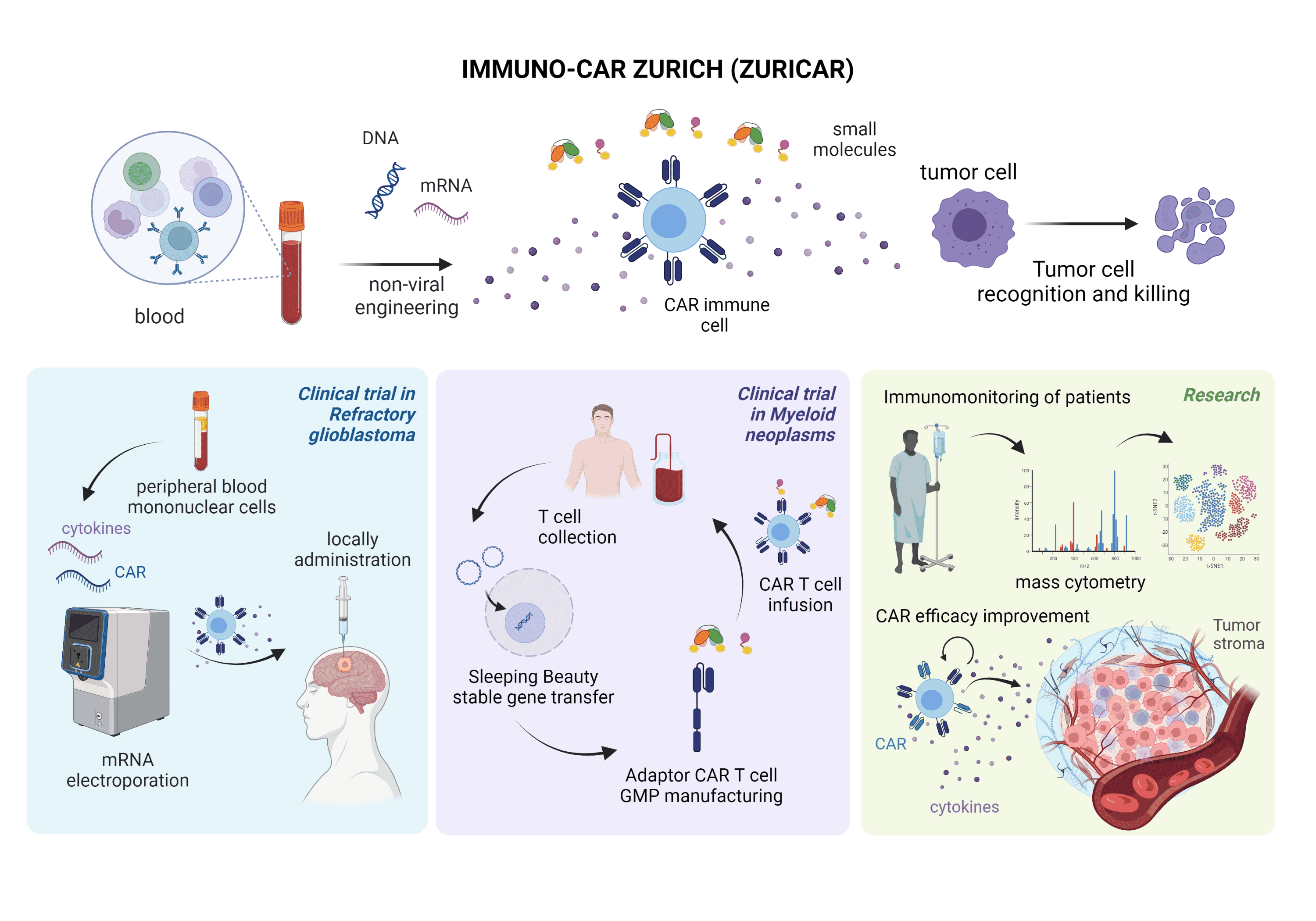
The first platform is based on highly efficient mRNA electroporation into non-activated, non-expanded peripheral blood mononuclear cells. This will enable a rapid clinical translation of innovative CAR products as it does not need cell activation or expansion and does not persistently change the genome of the cells. This platform will be applied within a phase I clinical trial to investigate the safety of local, intratumorally administered, multifunctional CAR PBMCs co-expressing a multi-targeting CAR and inflammatory cytokines in patients with refractory glioblastoma.
2: GMP production and clinical translation of Sleeping Beauty transposon engineered adaptor CAR T-cells
The second strategy is based on non-viral, switchable, multi-targeting adaptor CAR T-cells using the Sleeping Beauty transposon system in combination with small adaptor molecules. This platform will allow stable, cost-efficient transgene integration with increased safety based on on/off systems and increased efficacy by combinatorial targeting. CAR T cells will be manufactured in GMP by Wyss Zurich and will be evaluated pre-clinically before proceeding to a phase I/II trial. The first trial will be activated for patients with acute myeloid leukemia. Subsequently, we forseen application in solid tumors in combination with small molecules.
3: Research on CAR immunomonitoring and efficacy improvement
We will study the effect of CAR cell therapy using state-of-the-art technologies for immunomonitoring of patients including single-cell mass and flow cytometry of CAR immune cells and tumor ecosystems in samples from the clinical trials. Furthermore, we will pre-clinically explore strategies to boost Ad-CAR T cell efficacy by co-delivery of mRNA encoding for i) cytokines and ii) CAR molecules targeting the tumor stroma.
Overall, this project will address unmet needs for patients with difficult-to-treat malignancies and will spearhead innovative clinical CAR immune cell platforms at CCCZ.
“This project aims to develop and apply in the clinic innovative concepts of advanced medicine using gene therapy and cell transfer of CAR immune cells with increased efficacy, and safety for individuals with late-stage cancers who have limited other treatment options. Through the strict collaboration of scientists, doctors, and physician-scientists belonging to the Comprehensive Cancer Center, this project will facilitate the formation of a platform of cell therapies in Zurich and increase their applicability in oncology.”
Chiara F. Magnani - Dep. of Medical Oncology and Hematology
Tobias Weiss - Dep. of Neurology

Principal investigator(s) and departments

Prof. Dr. med. Chiara F Magnani, Wissenschaftliche Mitarbeiterin, University Hospital Zurich, Department of Medical Oncology and Hematology, chiara.magnani@usz.ch

PD Dr. med. Dr. sc. nat. Tobias Weiss, Leitender Oberarzt, University Hospital Zurich, Department of Neurology, tobias.weiss@usz.ch

Prof. Dr. med. Markus G. Manz, Medical Director and Chair CCCZ, University Hospital Zurich, Department of Medical Oncology and Hematology, markus.manz@usz.ch

Prof. Dr. Bernd Bodenmiller, Director CCCZ Technologies, University of Zurich, Department of Quantitative Biomedicine, bernd.bodenmiller@biol.ethz.ch
Funding Period 1.7.2023-31.12.2026
Funding Volume CHF 2.000.000
Funded by University Medicine Zurich (UMZH), Comprehensive Cancer Center Zurich (CCCZ)
Further projects
CCCZ Investigator Initiated Trial
BRAVE-Lung
Robotic-assisted Bronchoscopic Radiofrequency Ablation for Very-Early Lung Cancer -A novel Cancer Therapy
CCCZ Leuchtturmprojekte
Interdisziplinäre Forschungsteams übertragen Erkenntnisse aus der Grundlagenforschung in die klinische Anwendung und entwickeln neue Behandlungskonzepte gegen Krebs.