Keywords
Immunosuppressive Macrophages, Macrophage-Drug-Conjugates, Glioblastoma, Lymphoma, Myeloid Leukemia
Overarching Goal/Mission Statment
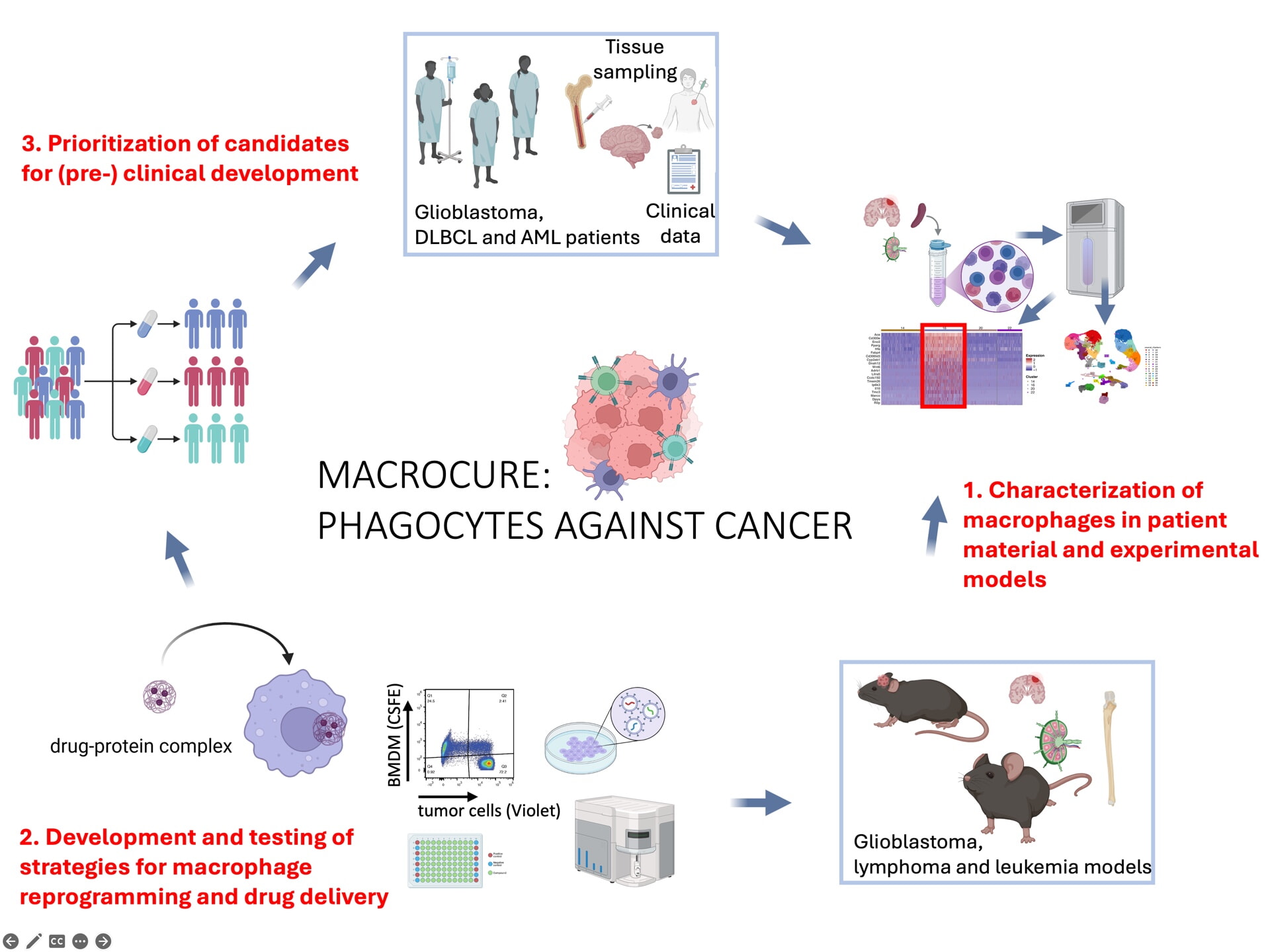
Our goal is to describe the immunophenotype and transcriptional signature of tumor-infiltrating macrophages in glioblastoma, diffuse large B-cell lymphoma and acute myeloid leukemia by applying single cell and high-resolution approaches to patient material and suitable mouse models. In a second step, macrophages will be targeted for reprogramming using mono- and bi-specific antibodies, and used as drug delivery platforms in preclinical models of the three malignancies; the final goal is to prioritize one or two strategies for clinical development.
Aims
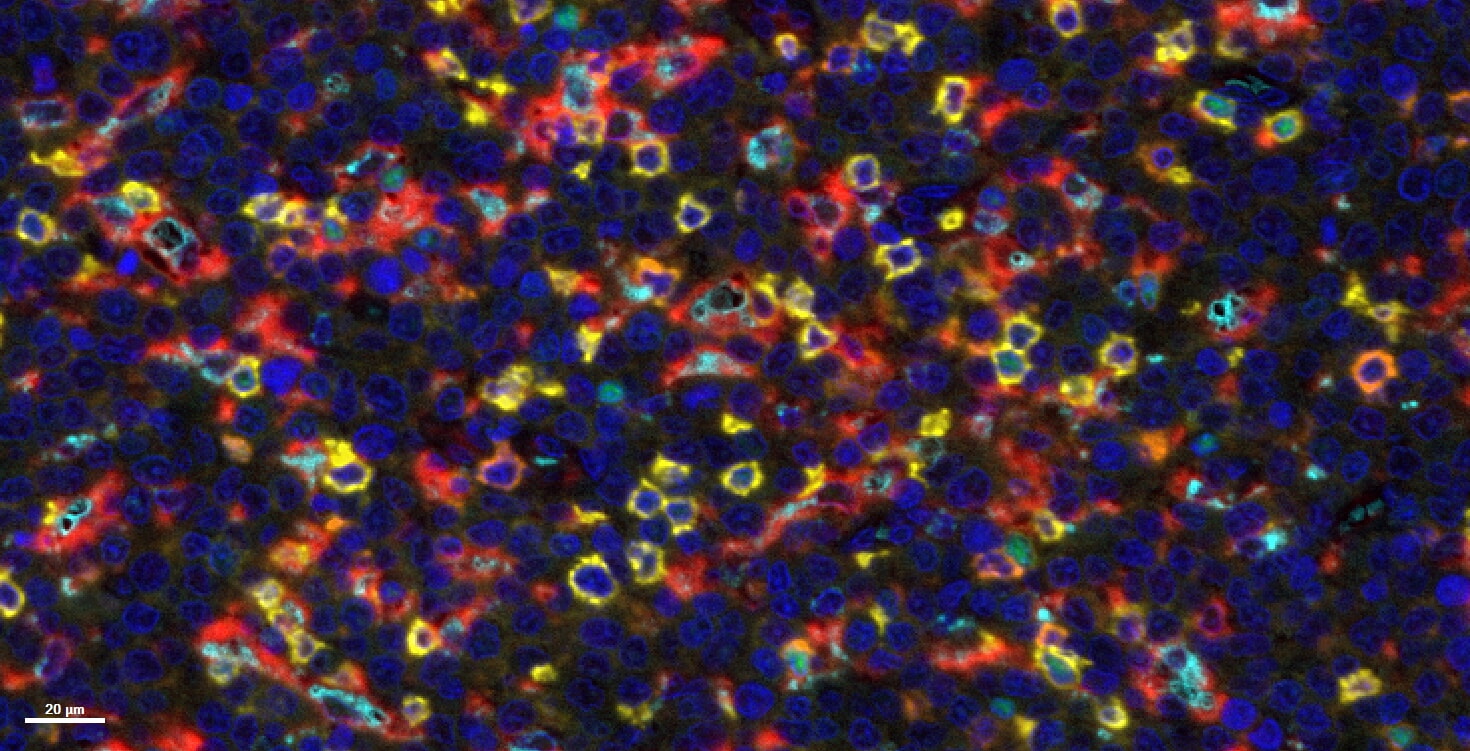
- Aim 1: Characterize macrophages within the tumor microenvironment of DLBCL, AML, and primary brain tumors using patient samples. This will involve the application of customized single-cell and spatial transcriptomic technologies to patient cohorts, with the goal being the description of a shared TAM signature across tumor entities or to understand the differences.
- Aim 2: Characterize macrophages in experimental models of DLBCL, AML, and primary brain tumors and investigate their pro-tumor activity using genetic tools and depleting antibodies. The signature capture will involve similar approaches as described for Aim 1; the functional studies in new transgenic mouse models will deliver proof-of-concept for a key role of TAM subsets in sustaining a tumor-promoting TME in all three tumor entities.
- Aim 3: Develop and evaluate innovative macrophage-targeting strategies including macrophage reprogramming by mono- and bi-specific antibodies and the use of macrophages as drug delivery platforms, and to prioritize one (maximum two) therapeutic candidates for preclinical development. To this end, we will leverage the models and insights generated in Aims 1 and 2 and will develop novel intervention strategies targeting macrophages; this aim will culminate in the development of a trial protocol towards the initiation of an investigator-initiated phase I clinical trial.
“Our mission is to better understand tumor-infiltrating macrophages in order to target them more precisely and harness them in the fight against cancer.”
Prof. Dr. Anne Müller - Institute of Molecular Cancer Research, UZH

Project Summary
Tumor-associated macrophages (TAMs) are a ubiquitous component of the tumor microenvironment, present in both immunologically “hot” and “cold”, i.e. adaptive immune cell infiltrated or non-infiltrated tumors. They can promote cancer growth and progression, yet they also hold the capacity to support tumor eradication, particularly in the context of immunotherapy. TAMs exist in diverse functional states and arise from distinct ontogenies, including blood-derived monocytes and embryonically seeded tissue-resident macrophages. Their transcriptional and functional signatures are strongly shaped by the tumor context and evolve over time. To fully exploit the therapeutic potential of cancer immunotherapy, we must extend our focus beyond adaptive immune cells and additionally harness the multifaceted power of macrophages. Our consortium combines leading immune-oncology PIs with expertise in macrophage biology and collaborators with specific pre-clinical and clinical expertise in the three paradigmatic malignancies that are at the center of this proposal, i.e. diffuse large B-cell lymphoma acute myeloid leukemia and glioblastoma. We share a common interest in better understanding the (patho)biology of TAMs for the purpose of therapeutically targeting these cells and utilizing their anti-cancer potential, either through reprogramming to induce anti-tumor responses or by harnessing their ability to deliver drug conjugates to tumor cells. We have established preclinical in vitro and in vivo syngeneic and humanized mouse models, which allow the investigation of novel macrophage-targeting strategies, and we have access to patient samples from large cohorts. Within Aim 1, we will take advantage of a variety of well-established single cell technologies (spectral flow cytometry, multiplex imaging and scRNA sequencing) and spatial transcriptomics to describe the signature and spatial context of TAMs in patient tumor material across the three tumor entities. In Aim 2, experimental syngeneic models of the three entities will be subjected to macrophage profiling using the technologies of Aim 1, and will further be genetically targeted for ablation of key putative immunomodulatory macrophage activities to deliver proof-of-concept for a functional role of macrophages in immunosuppressive/-evasive modulation of the tumor microenvironment. Within Aim 3, we will develop various complementary strategies of macrophage targeting that range from reprogramming and activating macrophages using mono- or bi-specific antibodies (e.g., blocking CD47-SIRPα, stimulation of CD40, and exploiting the targets identified in Aims 1 and 2) to harnessing monocyte-derived macrophages, which are attracted to tumors and engage with cancer cells, as drug-conjugate delivery platforms. We will test these strategies in our existing models, alone and in combination with standard-of-care or other (immuno)therapeutic approaches. Reagents will be shared and tested across entities. A final goal of Aim 3 is the selection of the top 1-2 most promising strategies (and entities) and the initiation of a preclinical development program, which will serve as the basis for a phase 1 clinical trial. Taken together, we will determine how specific TAM signatures translate into distinct functional subsets within the TME, how these signatures are conserved across tumor entities, and how they can be exploited for precise therapeutic targeting.
Principal Investigator and Project Team

Prof. Dr. Anne Müller, Director CCCZ Basic Research, University of Zurich, Institute of Molecular Cancer Research, mueller@imcr.uzh.ch

Prof. Dr. med. Markus G. Manz, Medical Director and Chair CCCZ, University Hospital Zurich, Department of Medical Oncology and Hematology, markus.manz@usz.ch

Prof. Dr. Melanie Greter, University of Zurich, Institute of Experimental Immunology, greter@immunology.uzh.ch

Prof. Dr. med. Alexandre Theocharides, Director CCCZ Clinical Trials Program, University Hospital Zurich, Department of Medical Oncology and Hematology, alexandre.theocharides@usz.ch

Prof. Dr. Burkhard Becher, University of Zurich, Institute of Experimental Immunology, becher@immunology.uzh.ch

PD Dr. med. Dr. sc. nat. Tobias Weiss, Leitender Oberarzt, University Hospital Zurich, Department of Neurology, tobias.weiss@usz.ch
Funding Period
1.1.2026-31.12.2029
Funding Volume
CHF 1.750.000
Funded by
University Medicine Zurich (UMZH)
Comprehensive Cancer Center Zurich (CCCZ)
Further projects
CCCZ Investigator Initiated Trial
BRAVE-Lung
Robotic-assisted Bronchoscopic Radiofrequency Ablation for Very-Early Lung Cancer -A novel Cancer Therapy
CCCZ Leuchtturmprojekte
Interdisziplinäre Forschungsteams übertragen Erkenntnisse aus der Grundlagenforschung in die klinische Anwendung und entwickeln neue Behandlungskonzepte gegen Krebs.